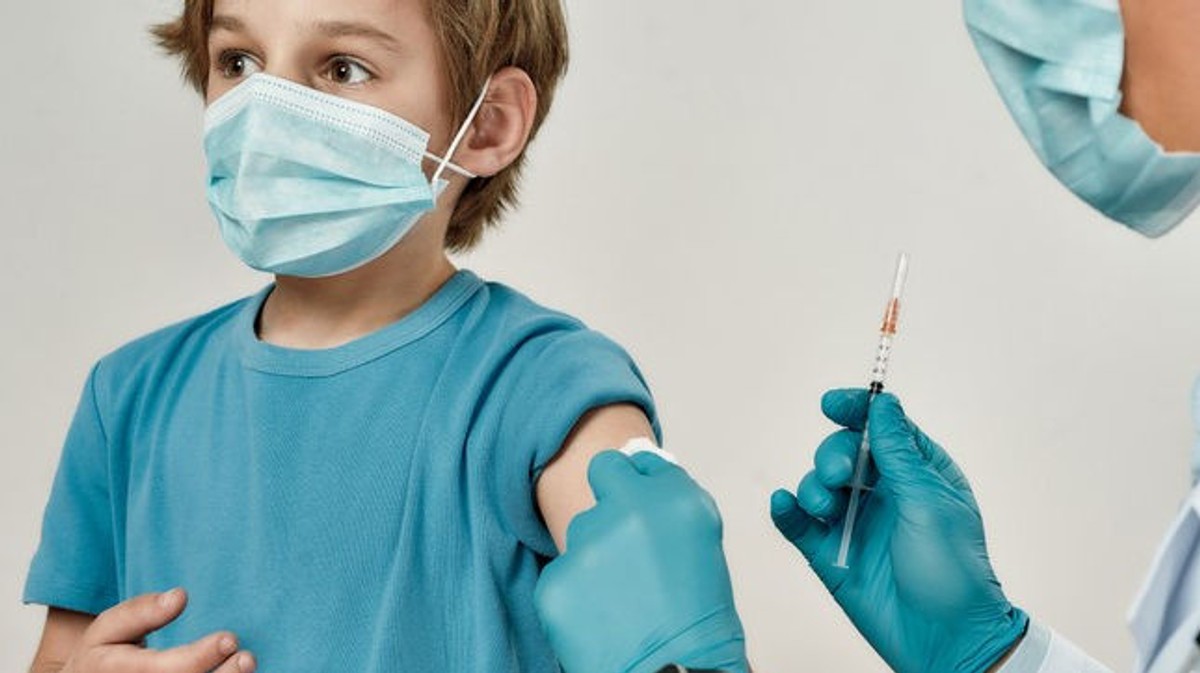
The Czech Republic shall take the necessary steps. Ministerstvo už objednalo vakcíny
The Czech Republic may, in particular, endorse the coronavir and apply it only after 5 to 11 years. The Ministry of Transport has, for the time being, 140 years of service, which must be approved by the Member States. In addition, Pfizer and BioNtech have agreed that the vaccine will be used in this category.
In addition, Pfizer and BioNTech have been able to provide vaccines for their studies, for which a total of 2268 decisions were taken between 5 and 11 years. “A total of 10 micrograms of mNRA vaccine should be applied to each day.
The European Commission of the European Agency for Reconstruction and Development (EMA) is responsible for implementing the European Central Audit Office (EMA). “It is therefore necessary for this Regulation to apply to the Commission and to the Commission for the registration, so that it can be extended to all the EU Member States and not to any other Member State,”
In addition, the EMA may, if necessary, for the purposes of this Regulation, determine whether the vaccine has been registered in accordance with that Regulation.
“My mission is to provide the company with Pfizer, and we are responding to the issue of vaccines in the Czech Republic.”
This is a practical practice
The Czech Republic has an object of 140 years. It is necessary to use practical measures to ensure that the rules are in place. “Budeme-li chtít očkovat děti mladší 12 let, tak budeme mít předpřipravené dávky právě pro tuto categorie ni nišší concentrací té úcinné složky vaccine”, přiblížila pediatric Hana Cabrnochová.
“If it is necessary to do so, the vaccine should be used. It is recommended that the vaccine be classified in one of the following categories and that it should be used without further treatment.”
If this is not possible, the vaccine will not be available for all 12 years. In the case of a group of 12 to 15 years, the vaccine for which the vaccine was granted in the Czech Republic for a period of 145 years shall not exceed three months.
Do you want to know how to use it? Report report:
ls, TN.cz