Mystery of the active phase in catalytic carbon dioxide reduction solved – University of Innsbruck
The measurements for the present work were carried out on the PETRA III beamline P22 at the German Electron Synchrotron DESY. In the picture the PETRA III experimental hall to an early stadium.
An international research team led by Stockholm University and with the participation of Bernhard Klötzer and his working group from the Innsbruck Institute for Physical Chemistry has for the first time succeeded in observing the surface of a copper-zinc catalyst in the reduction of carbon dioxide to methanol “live”. The results were published in the journal Science.
Methanol is currently one of the most important petrochemical raw materials with an annual production of 110 million tons. It can be turned into tens of thousands of different products and used to make plastics, detergents, medicines and fuel. Methanol also has the potential to become a future non-fossil fuel, for example by making jet fuel from recycled carbon dioxide and hydrogen from the electrolysis of water with renewable electrical energy. The technical production of methanol takes place in a catalytic process in which a mixture of carbon monoxide (CO), carbon dioxide (CO2) and hydrogen (H2) reacts to form methanol (CH3OH). The technical catalyst is made up of copper, zinc oxide and aluminum oxide. However, the underlying function and the active chemical state of the catalyst components during the technical synthesis reaction have not yet been clarified.
in situ experiment
The aim of the present work was therefore to experimentally examine the catalyst surface in its active state, i.e. “live”, using surface-sensitive methods under relevant pressure and temperature conditions. These conditions were unattainable for many years, and there have been broader hypotheses as to whether zinc exists primarily as the oxide, metallic, or in an alloy with copper. So far, none of these hypotheses or the model calculations based on them have been clearly verified.
The in situ experiments were performed at the PETRA III beamline P22 at the Deutschesuron-Synchrotron DESY, which is optimized for studying the electronic and atomic structure and chemical state of surfaces, interfaces and bulk materials. To this end, the scientists at Stockholm University have developed an in situ photoelectron spectroscopy instrument that can be used to examine the catalyst surface at previously unattainable sampling rates, allowing them to directly observe what is happening in the reaction close to technological conditions.
Bernhard Klötzer’s “Nanostructured model catalysis” working group at the Institute for Physical Chemistry at the University of Innsbruck has developed substantial expertise in the past few years in understanding how catalysts work at the atomic level. Her methodological approach, in which she examines model catalysts that are particularly suitable for surface spectroscopy during the ongoing catalysis process (“in-situ”), plays a particularly important role. “In the current work, we were able to use this many years of experience to create a realistic model surface that simulates the active centers of the industrial copper-zinc catalyst,” explains Bernhard Klötzer.
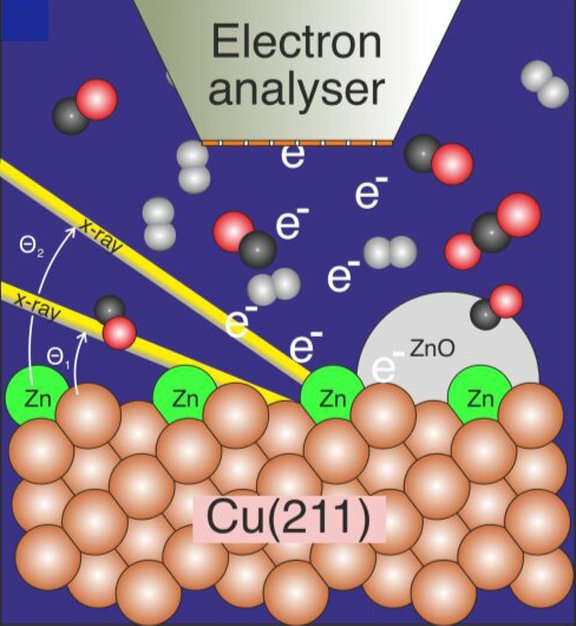
The graphic shows a copper surface of the catalyst covered with a small amount of zinc, which is examined using photoelectron spectroscopy during the reaction of carbon dioxide, carbon monoxide and hydrogen to form methanol.
Together, the researchers were able to prove that “active” zinc is alloyed with copper directly on the metal surface, resulting in bimetallic active centers where methanol is formed from carbon dioxide particularly efficiently. In addition, they were able to determine different active states of the catalyst surface depending on the CO and CO2 content in the gas mixture – a starting point for optimizing existing processes or even for searching for new, better catalysts.
The study was carried out by the University of Stockholm in cooperation with the University of Innsbruck and the Technical University of Vienna, DESY and the Fritz Haber Institute in Berlin.




