Large study of side effects against COVID vaccine in Sweden
With mass vaccination against COVID-19 worldwide in December 2020, regulators, academia, pharmaceutical companies and other stakeholders are working on different pharmacovigilance strategies based on observational data.
The vACCine covid-19 monitoring readinESS (ACCESS) project in Europe is funded by the European Medicines Agency and contains a protocol for evaluating background rates for conditions that represent potential advers evalves off sspecial ininterest (AESI) from observational data.
Studying an age- and gender-stricken Swedish cohort for AESI results through the COVID-19 pandemic
Recently, researchers from Sweden reported population-based, age- and gender-specific background incidents of conditions that represent potential COVID-19 vaccine side effects of particular interest (AESI) in the general Swedish population using registered data. This study is published on medRxiv* oppression server.
The researchers analyzed an age- and gender-stratified, random 10% sample of the Swedish population on 1 January 2020, followed up a year for AESI results through the COVID-19 pandemic, before vaccinations were approved or started.
The following results were selected and defined by the authors based on information from regulators, previous studies, large-scale initiatives: febrile seizures, aseptic meningitis, Kawasaki syndrome, arthritis, post-infectious arthritis, myocarditis, myocardial infarction, ARDS, stroke, hemorrhagic stroke, ischemic stroke, venous thromboembolism, renal failure, pulmonary embolism, liver failure, `erythema multiforme, autoimmune thyroiditis, disseminated intravascular coagulation and appendicitis.
The determined incidence rates stratified by age, sex, and time period and classified them according to the following Council of International Organizations of Medical Sciences (CIOMS) categories: very common, common, unusual, rare, or very rare.
The incidence of side effects varied greatly by age and in some cases gender
A total of 972,723 subjects who represented the Swedish population on 1 January 2020 were included in the study. They found that the prevalence of AESI varied significantly by age and in some cases by gender. Many common AESIs increased, as expected, with age, while some AESIs, such as appendicitis, autoimmune thyroiditis, aseptic meningitis, Kawasaki syndrome, and MISC, were more common among young people. On the other hand, other AESIs such as myocarditis and erythema multiforme showed a flatter age pattern.
As a result, the CIOMS rating for AESIs varied greatly depending on age. The results showed great variation over the four quarters of 2020 for certain AESI prices, which may be related to pandemic waves, congestion in the healthcare system, other health effects or seasonal variations. In summary, age, gender, and timing of rates are important variables to consider when comparing background AESI rates with similar frequencies observed with COVID-19 vaccines.
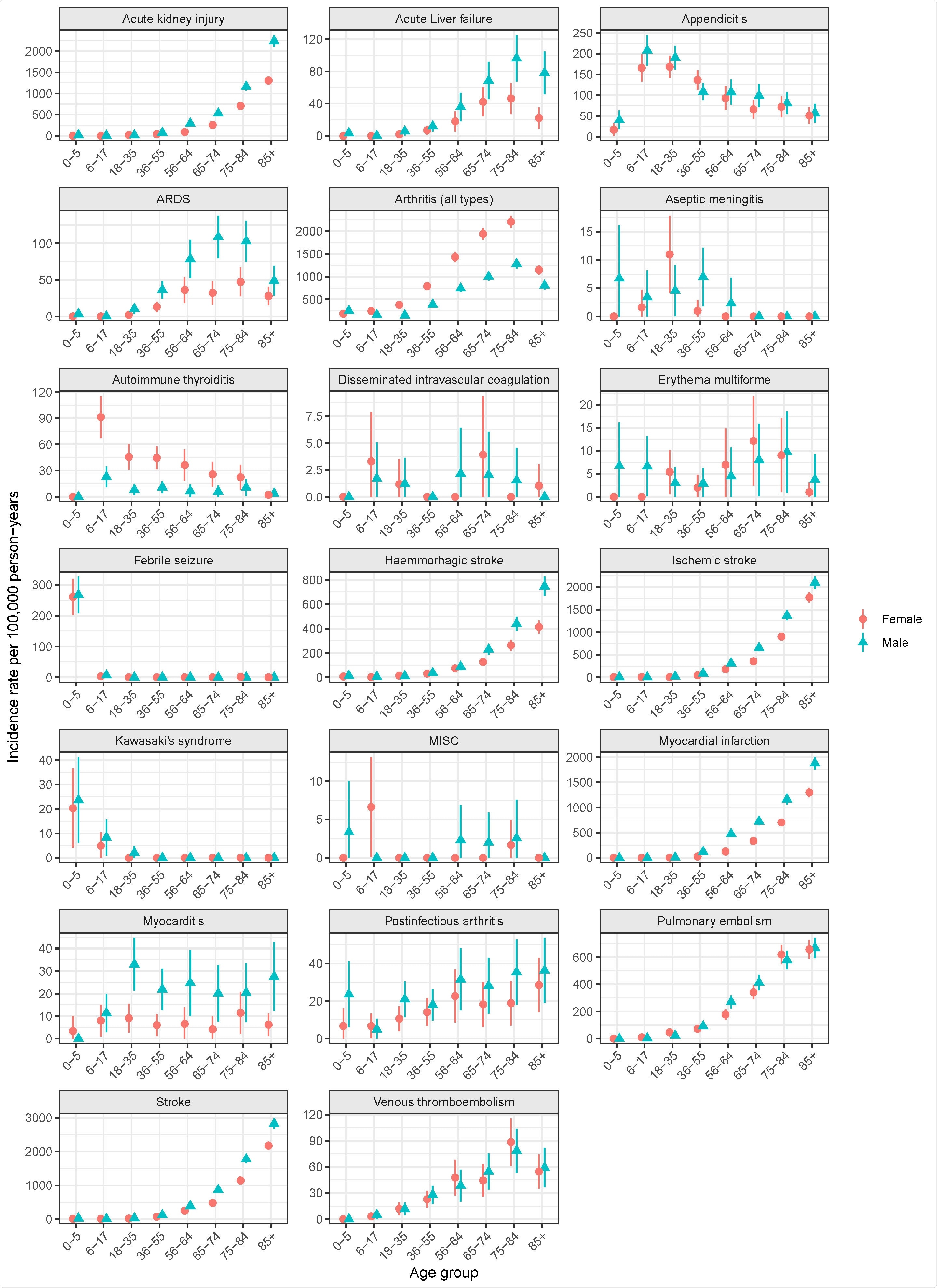
Age- and gender-stratified incidence of 14 adverse reactions of special interest (AESI) per 100,000 persons (with a 95% confidence interval) in the Swedish population
Demographic comparability and population heterogeneity must be taken into account when evaluating AESI
This study reported descriptive epidemiology of many potential AESIs for the safety follow-up of COVID-19 vaccines from a random Swedish population. The work described AESI’s baselines in 2020 as a whole and per quarter, and the results show some crucial points for future vaccine safety studies.
“The clear quarterly trends seen for some AESIs during the pandemic in Sweden, with varying patterns, mean that it is crucial to compare prices over appropriate time periods.”
According to the authors, the significant variation in rates by age and for some results even by gender indicates the importance of sufficient demographic comparability for vaccinated and non-vaccinated groups compared. It is also important to ensure adequate adaptation or standardization for age and gender while using background speeds for security monitoring.
Recent major international studies have shown significant heterogeneity between populations; For example, data from the ACCESS project showed similar quantities of heterogeneity in background frequencies. The OHDSI group study reported variations between databases and websites. This emphasizes the fact that the heterogeneity of the population must be taken into account when evaluating AESI.
“These background frequencies provide useful real-world clinical contexts for vaccine monitoring activities and ensure patient safety as Covid-19 vaccines are used to combat pandemics worldwide.”
*Important message
medRxiv publishes preliminary scientific reports that have not been peer-reviewed and therefore should not be considered as decisive, guide clinical practice / health-related behavior or be treated as established information.
Journal reference:
- Side effects of particular interest to COVID -19 vaccines – background events vary depending on gender, age and time period and are affected by the pandemic Fredrik Nyberg, Magnus Lindh, Lowie EGW Vanfleteren, Niklas Hammar, Björn Wettermark, Johan Sundström, Ailiana Santosa, Brian K Kirui, Magnus Gisslén, medRxiv, 2021.10.04.21263507; doi: https://doi.org/10.1101/2021.10.04.21263507, https://www.medrxiv.org/content/10.1101/2021.10.04.21263507v1

.jpg)


